トラック&トレースソリューション市場の成長を牽引する主な要因には、医療機器および製薬業界の成長、シリアライゼーションの実施の増加、包装関連製品のリコール数の増加、偽造医薬品の増加などがあります
大規模な製薬会社やバイオ製薬会社が直面する大きな問題の1つは、医薬品の偽造です

トラック&トレースソリューション市場の成長を牽引する主な要因には、医療機器および製薬業界の成長、シリアライゼーションの実施の増加、包装関連製品のリコール数の増加、偽造医薬品の増加などがあります
大規模な製薬会社やバイオ製薬会社が直面する大きな問題の1つは、医薬品の偽造です。ブランドがブランド認知度を確実に得るために、これらの企業はサプライチェーン監視にトラック&トレースソリューションの使用を採用しています。偽造はさまざまな方法で発生します。それは、本物の承認された製品を複製する目的で薬を誤って表示することから、有効成分なしで薬を販売したり、有効成分の不十分または過剰な量を追加したりするはるかに危険な慣行まで、多岐にわたります。偽造医薬品には、無関係または有害な化学物質が含まれていることもあります。トラック&トレースソリューションの出現により、この問題は非常に大きく根絶されることが期待されています。その結果、この要因は、製薬会社の間での採用の増加を促進し、予測期間中の市場の成長をさらに推進しています。ただし、シリアル化と集約に関連する高いコストや、シリアル化と集約に関する共通の標準の欠如などの要因が、市場の成長を妨げる可能性があります
主な市場動向
バーコード技術は、技術面でのセグメンテーションで主要な収益シェアを保持すると予想されています
バーコードは、さまざまな太さの縦線で表され、ラベルに印刷された数値で、アイテムを排他的に識別します。
ヘルスケア業界では、バーコードシステムは、病院や診断研究所から医療機器や製薬製造会社まで、幅広いアプリケーションを提供します。バーコードは、医療施設内の患者データの追跡、医療用品の在庫の正確な管理、医薬品の有効期限と原産地の追跡、血液供給へのバーコードラベルの追加など、さまざまなサンプルを区別するのに役立ちます。このバーコード技術は長年にわたって採用されており、現在でも追跡に常に使用されています。バーコードを使用する他の主な利点は、在庫の可視性の向上と無駄の削減、コストとケアの質の格差の削減、安全性とコンプライアンスの向上、手作業によるサプライチェーンタスクの自動化であり、これらは市場の成長を促進する主な推進要因です
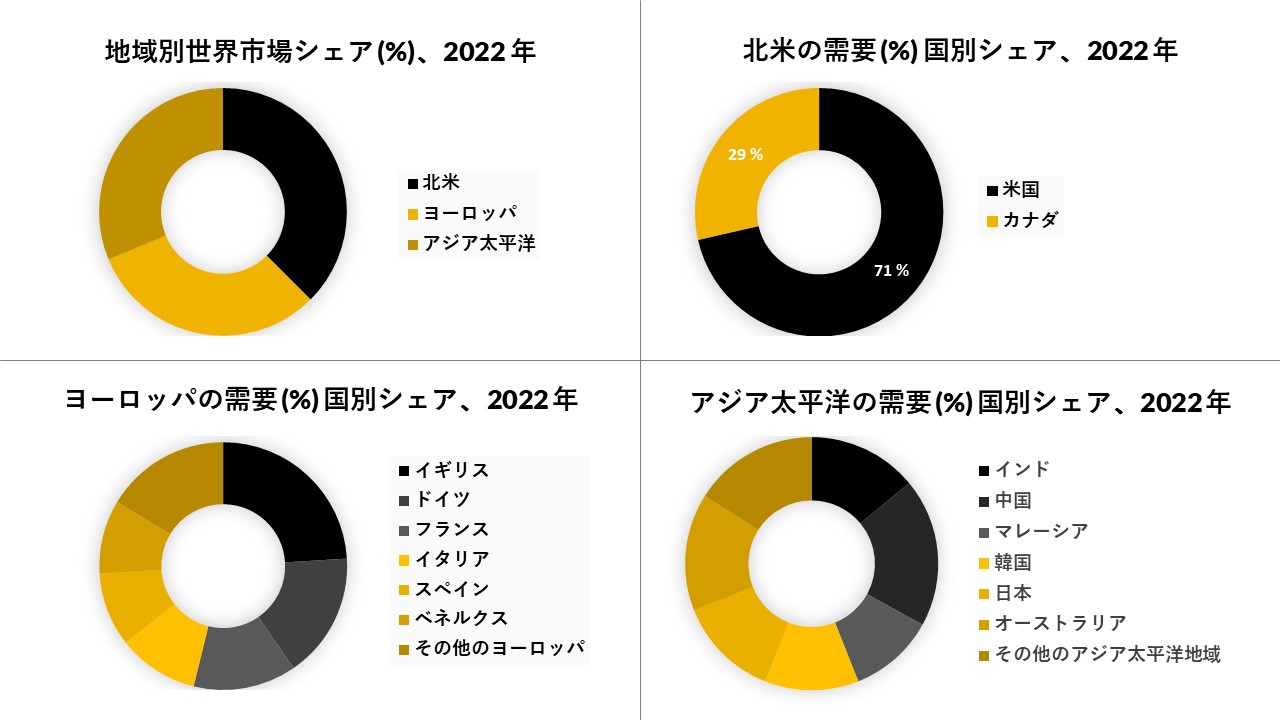
北米が市場を支配しており、予測期間
北米は現在、トラック&トレースソリューションの市場を支配しており、今後数年間は拠点を継続すると予想されています。北米地域では、米国が最大の市場シェアを占めています。これは主に偽造医薬品の数が増えていることに起因しています。市場で入手可能な偽造医薬品の数が増えているため、FDAは2017年6月に医薬品サプライチェーンセキュリティ法(DSCSA)コンプライアンスポリシーに基づいて製品識別子の要件を発行しなければなりませんでした。このポリシーでは、米国で販売される医薬品には、販売単位の処方薬に品目レベルのシリアル化が貼付され、印刷されている必要があります。DSCSAは、国家医薬品コード(NDC)によって構成されるパッケージまたは均質な症例を一意に識別するために、「標準化された数値識別子」を必要とします。DSCSAは、国家医薬品コード(NDC)によって構成されるパッケージまたは均質な症例を一意に識別するために、「標準化された数値識別子」を必要とします><。
競争環境
トラック&トレースソリューション市場は競争が激しく、いくつかの主要プレーヤーで構成されています。市場シェアの面では、現在市場を支配している主要プレーヤーはほとんどありません。トラック&トレースソリューションに対する意識の高まりと医療インフラの改善に伴い、市場に参入する小規模なプレーヤーはほとんど期待されていません。市場の主要プレーヤーのいくつかは、とりわけAxway、Mettler-Toledo International Inc.、Rfxcel Corporation、Sea vision SRL、TraceLink Inc.です><。
このレポートを購入する理由:
- エクセル形式の市場予測(ME)シート
- 3ヶ月のアナリストサポート

北米(米国およびカナダ)、ラテンアメリカ(ブラジル、メキシコ、アルゼンチン、その他のラテンアメリカ)、ヨーロッパ(英国、ドイツ、フランス、イタリア、スペイン、ハンガリー、ベルギー、オランダおよびルクセンブルグ、NORDIC(フィンランド、スウェーデン、ノルウェー) 、デンマーク)、アイルランド、スイス、オーストリア、ポーランド、トルコ、ロシア、その他のヨーロッパ)、ポーランド、トルコ、ロシア、その他のヨーロッパ)、アジア太平洋(中国、インド、日本、韓国、シンガポール、インドネシア、マレーシア) 、オーストラリア、ニュージーランド、その他のアジア太平洋地域)、中東およびアフリカ(イスラエル、GCC(サウジアラビア、UAE、バーレーン、クウェート、カタール、オマーン)、北アフリカ、南アフリカ、その他の中東およびアフリカ)