肝吸虫治療市場は、予測期間中に4.2%のCAGRを登録すると予想されています

肝吸虫治療市場は、予測期間中に4.2%のCAGRを登録すると予想されています。肝吸虫は、ヒト、ウシ、ヒツジに筋膜症と呼ばれる病気を引き起こす可能性のある寄生虫です。肝吸虫感染は、Fasciola hepaticaやFasciola giganticaなどのワームによって引き起こされます。ヒトの感染は、通常、汚染された生または調理不足の淡水魚またはクレソンを食べた後に起こります。肝吸虫感染者は、ほとんど何の症状も示さず、時には胆道系に関連する症状が現れることがあります。まれに、長期的な合併症も発症する可能性があります。肝吸虫感染の兆候のいくつかは、腹痛、倦怠感、下痢、吐き気と嘔吐、発熱、食欲減退と体重減少です.
疾病管理予防センター(CDC)によると、筋膜症は世界の多くの地域で発生しており、羊や牛の一般的な肝臓吸虫です。さらに、筋膜症は人よりも動物でより一般的で広まっています。それでも、感染者数は世界で200万人を超えると考えられています。さらに、The American Journal of Tropical Medicine and Hygieneに掲載された研究によると、世界中で260万人の感染者と9100万人の感染リスクがあると推定されています.
ヒトおよび動物における肝吸虫感染の症例の増加および人口における肝吸虫感染に対する意識の高まりは、肝吸虫治療市場における主要な推進要因である
主な市場動向
トリクラベンダゾールセグメントは、肝吸虫治療市場で主要な市場シェアを保持すると予想されています
- トリクラベンダゾールは、筋膜症の治療のための獣医の練習で日常的に使用されるベンズイミダゾール化合物です。1997年、筋膜症の治療にトリクラベンダゾールを使用した臨床試験の後、世界保健機関(WHO)は、この薬を必須薬リストに載せることを推奨しました.
世界保健機関(WHO)によると、世界中で70カ国以上で少なくとも240万人が筋膜症に感染し、数百万人が危険にさらされていると推定されています。さらに、牛や羊が飼育されているほぼすべての国で家畜に感染が存在します
- トリクラベンダゾールセグメントは、肝吸虫治療市場で大きな市場シェアを保持しており、予測期間中に同様の傾向を示すと予想されています
- さらに、トリクラベンダゾールは他の薬剤と比較して非常に効果的であり、疾病管理センター(CDC)および世界保健機関(WHO)によって肝吸虫感染症に対する第一選択療法と考えられている.
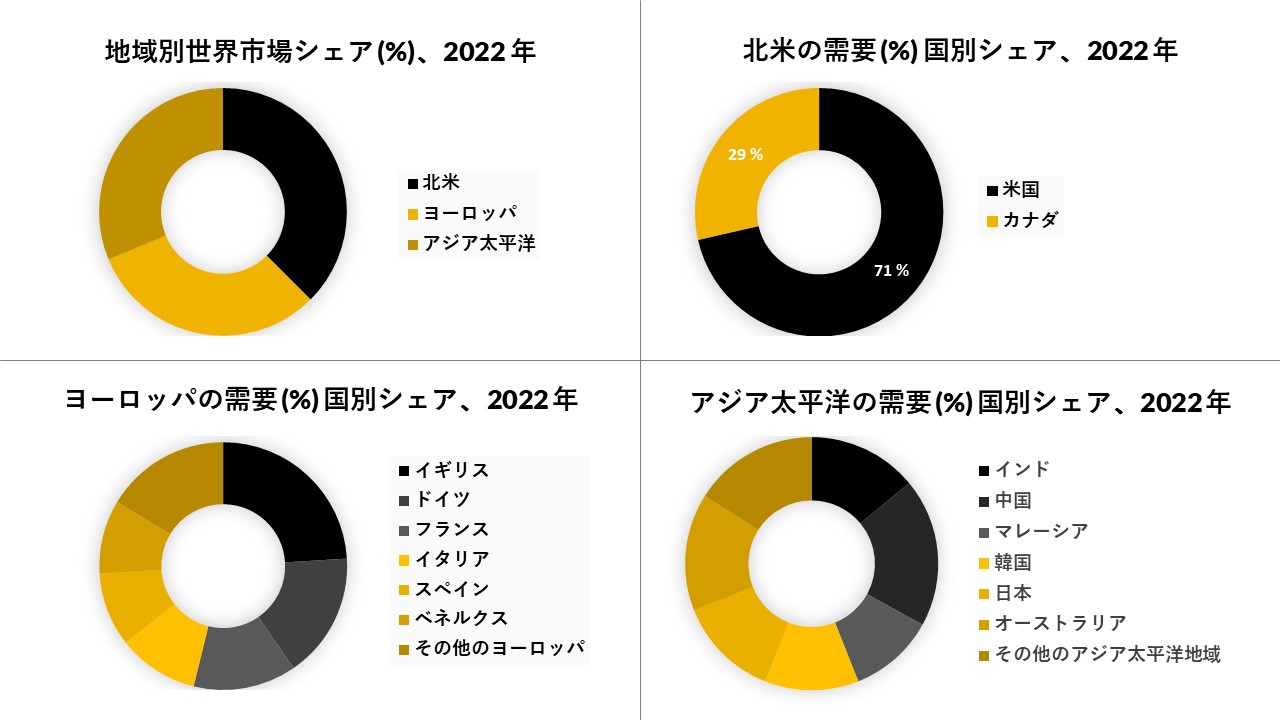
北米は市場で大きなシェアを占めると予想され、予測期間
北米は、この地域の牛や羊における筋膜肝感染症の有病率の増加により、世界の肝吸虫治療市場で大きな市場シェアを保持すると予想されています。The American Journal of Tropical Medicine and Hygieneに掲載された研究によると、獣医学的疾患は米国の複数の地域で流行しており、おそらく拡大していると推定されています。さらに、カリフォルニアの牛における筋膜肝感染症の有病率が高く、一部の地域では、成牛の90%が筋膜肝に感染しています。さらに、肝吸虫治療の研究開発の増加も、地域市場全体の成長を促進しています
競争環境
肝吸虫治療市場は競争が激しく、いくつかの主要なプレーヤーで構成されています。ノバルティスAG、グラクソ・スミスクライン、バイエルAG(バイエル・アニマルヘルス)、遠藤インターナショナル(パー・ファーマシューティカル)、ロマーク・ラボラトリーズ、L.C、ゾーティス・インク、メルク・アンド・カンパニー、ルパン・ファーマシューティカルズ、シェリング・プラウ・コーポレーションなど、主要な市場プレーヤーが名を連ねています
このレポートを購入する理由:
- エクセル形式の市場予測(ME)シート
- 3ヶ月のアナリストサポート

北米(米国およびカナダ)、ラテンアメリカ(ブラジル、メキシコ、アルゼンチン、その他のラテンアメリカ)、ヨーロッパ(英国、ドイツ、フランス、イタリア、スペイン、ハンガリー、ベルギー、オランダおよびルクセンブルグ、NORDIC(フィンランド、スウェーデン、ノルウェー) 、デンマーク)、アイルランド、スイス、オーストリア、ポーランド、トルコ、ロシア、その他のヨーロッパ)、ポーランド、トルコ、ロシア、その他のヨーロッパ)、アジア太平洋(中国、インド、日本、韓国、シンガポール、インドネシア、マレーシア) 、オーストラリア、ニュージーランド、その他のアジア太平洋地域)、中東およびアフリカ(イスラエル、GCC(サウジアラビア、UAE、バーレーン、クウェート、カタール、オマーン)、北アフリカ、南アフリカ、その他の中東およびアフリカ)