凍結手術装置市場は、心血管疾患や皮膚疾患などの障害の増加により、成長率の増加を記録すると予想されます

凍結手術装置市場は、心血管疾患や皮膚疾患などの障害の増加により、成長率の増加を記録すると予想されます。世界保健機関(WHO)によると、心血管疾患は主要な死因であり、世界中で約1790万人が死亡しています
- さらに、Health and Safety Executiveのデータによると、2018年には、英国で約67人の新しい症例が仕事関連の皮膚がんで報告されていました。したがって、これはこれらの疾患の治療の必要性を浮上させる
●また、凍結手術装置の開発も市場を後押しすることが期待されています。2019年、CSA Medical Inc.は、その製品「若返りエアシステム」を販売する欧州の承認を受けました。これは、気管支鏡手術による慢性気管支炎の治療のためのメトレッドクライオススプレー(液体窒素)を含む凍結手術装置です。したがって、これらの要因は、凍結手術装置市場の成長を促進すると予想されます
主な市場動向
腫瘍障害セグメントは、凍結手術装置市場で大きなシェアを保持すると予想されています
腫瘍学セグメントは、がんの罹患率が高く、低侵襲手術に対する嗜好の高まりにより、凍結手術装置市場で大きなシェアを占めると予想されています。世界保健機関(WHO)によると、2018年のがんの負担は世界で約1810万件でした。これは世界で2番目に多い死因であり、上記の情報源によると約960万人の死亡を引き起こしています
さらに、凍結アブレーションは、経皮アブレーション法に亘って乳癌の治療に非常に有効であることが観察されている。世界保健機関(WHO)によると、2018年には肺がんと乳がんが最も多く、約210万人が診断されました。したがって、前述の要因は、凍結手術装置市場にプラスの影響を与えると予想されます
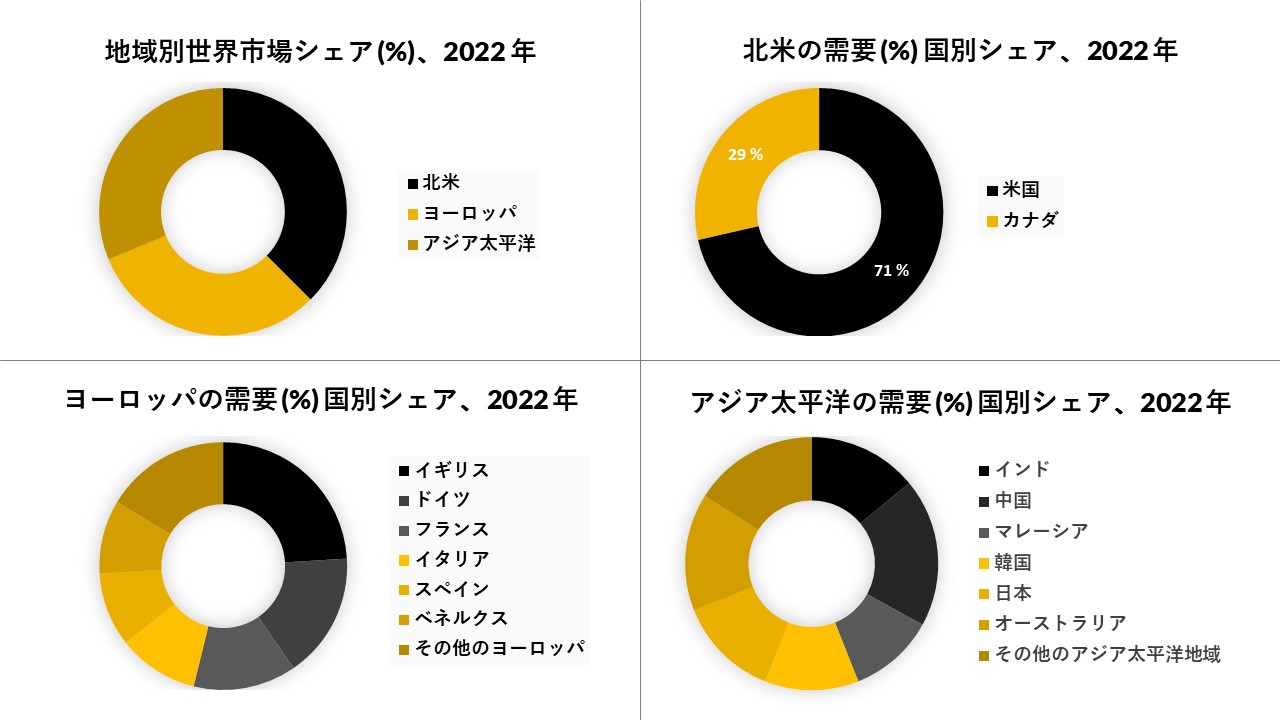
北米は市場で大きなシェアを占めると予想され、予測期間
北米は、慢性疾患の負担の増加と製品承認の存在により、調査対象市場にとって支配的な地域になると予想されています。国立がん研究所によると、2018年に米国で約174万人ががんと診断されました。さらに、2019年、米国FDAは、Cryoconceptsが開発した2つの凍結手術装置「CryoTouch」と「CryoLab」を承認しました。クライオタッチは、クライオラボが大量の手術に関与している間に、標的病変に低温剤を送達するポータブルデバイスです。これらは23の適応症に対して承認されています。したがって、このような要因が北米の凍結手術市場を牽引すると予想されています
競争環境
現在市場を支配している企業のいくつかは、ハーバードバイオサイエンス社、AtriCure Inc.、Brymill Cryogenic Systems、Cardinal Health、CryoConcepts、Cortex Technology、Bovie、Cooper Surgical.
このレポートを購入する理由:
- エクセル形式の市場予測(ME)シート
- 3ヶ月のアナリストサポート

北米(米国およびカナダ)、ラテンアメリカ(ブラジル、メキシコ、アルゼンチン、その他のラテンアメリカ)、ヨーロッパ(英国、ドイツ、フランス、イタリア、スペイン、ハンガリー、ベルギー、オランダおよびルクセンブルグ、NORDIC(フィンランド、スウェーデン、ノルウェー) 、デンマーク)、アイルランド、スイス、オーストリア、ポーランド、トルコ、ロシア、その他のヨーロッパ)、ポーランド、トルコ、ロシア、その他のヨーロッパ)、アジア太平洋(中国、インド、日本、韓国、シンガポール、インドネシア、マレーシア) 、オーストラリア、ニュージーランド、その他のアジア太平洋地域)、中東およびアフリカ(イスラエル、GCC(サウジアラビア、UAE、バーレーン、クウェート、カタール、オマーン)、北アフリカ、南アフリカ、その他の中東およびアフリカ)