
- 2020ー2024年
- 2026-2035年
- 必要に応じて日本語レポートが入手可能
自動液体ハンドラー市場エグゼクティブサマリ
1) 自動液体ハンドラー市場規模
ハンドラー市場に関する弊社の調査レポートによると、市場は予測期間2026―2035年において複利年間成長率(CAGR)7.2%で成長すると予想されています。来年には、市場規模は72.5億米ドルに達すると見込まれています。
しかし、弊社の調査アナリストによると、基準年の市場規模は32.9億米ドルに達しました。ここでの成長の原動力となっているのは、バイオ医薬品分野におけるイノベーションと臨床診断における自動化があります。

2) 自動液体ハンドラー市場の動向 – 好調な推移を示す分野
SDKI Analyticsの専門家によると、予測期間中に予測される自動液体ハンドラー市場の傾向には、創薬、臨床診断、ゲノミクス、プロテオミクス、学術研究などの分野が含まれます。予測期間中に自動液体ハンドラー市場を牽引すると予想される主要な傾向について、以下に詳細をご紹介します。
|
市場セグメント |
主要地域 |
CAGR(2026―2035年) |
主な成長要因 |
|
創薬 |
北米 |
7.2% |
AIベースのスクリーニング、医薬品研究開発の拡大、精密医療 |
|
臨床診断 |
ヨーロッパ |
6.8% |
高齢化、ラボの自動化、規制遵守 |
|
ゲノミクス |
アジア太平洋地域 |
7.1% |
集団規模のシーケンシング、バイオ技術への資金提供、技術導入 |
|
プロテオミクス |
北米 |
6.7% |
がんバイオマーカー研究、自動化、データ分析 |
|
学術研究 |
ヨーロッパ |
6.5% |
大学助成金、オープンサイエンス、共同プラットフォーム |
ソース: SDKI Analytics 専門家分析
3) 市場定義 – 自動液体ハンドラーとは何ですか?
自動液体ハンドラーとは、複数の研究プロジェクト、診断、さらには製薬アプリケーションにおいて、液体を効率的に分注、移送、混合するために設計された実験室機器を指します。プログラム可能なソフトウェアを搭載したロボットアームを使用することで、これらのシステムは人為的ミスの可能性を低減し、スループットを向上させ、サンプル調製、試薬分配、アッセイセットアップの精度を確保します。
4) 日本における自動液体ハンドラー市場規模:
日本の自動液体ハンドラー市場は大きな市場シェアを占めており、着実に成長しています。SDKI Analyticsの調査によると、予測期間中、市場は7.4%のCAGRで成長する見込みです。
日本における人口の高齢化、デジタルインフラの整備、そして医療改革を支援する政府の政策が、市場の成功を促進しています。2023年時点で65歳以上の高齢者人口は36.23百万人に達しており、自動診断の需要を牽引しています。さらに、調査によると、日本はAIの積極的な導入において世界第23位にランクされており、これも市場の成功を後押ししています。
- 日本の現地市場プレーヤーの収益機会:
日本の現地市場プレーヤーにとって、自動液体ハンドラー市場に関連するさまざまな収益機会は次のとおりです。
|
収益創出の機会 |
主要成功指標 |
主な成長要因 |
市場洞察 |
競争の激しさ |
|
ハイスループットスクリーニングソリューション |
採用率、ワークフロー効率 |
創薬需要、研究開発の拡大、ラボ自動化の動向 |
日本の製薬会社は、化合物のスクリーニングを加速するために自動化を導入する傾向が高まっています。 |
高い |
|
PCRセットアップ自動化 |
精度指標、セットアップ速度 |
感染症検査、遺伝子診断、検査標準化 |
臨床検査室では、PCR ワークフローの精度とスピードを重視し、自動化の導入を推進しています。 |
中 |
|
細胞培養液の取り扱い |
汚染管理、再現性 |
再生医療、幹細胞研究、バイオ技術への資金提供 |
日本では再生療法に重点が置かれており、無菌かつ一貫した液体処理の需要が高まっています。 |
中 |
|
学術研究の統合 |
Mask |
|||
|
開発業務受託機関(CRO)の需要 |
||||
|
ゲノム増幅ワークステーション |
||||
|
使い捨てチップシステム |
||||
|
マルチ機器統合プラットフォーム |
||||
ソース: SDKI Analytics 専門家分析
- 日本における自動液体ハンドラー市場の都道府県別内訳:
、日本における自動液体ハンドラー市場の都道府県別の内訳の概要です。
|
県 |
CAGR(%) |
主な成長要因 |
|
東京 |
7.4% |
高齢化、バイオ技術クラスター、AI統合 |
|
大阪 |
6.9% |
大学の研究拠点、製薬企業の拠点、自動化インフラ |
|
神奈川 |
Mask |
|
|
愛知 |
||
|
福岡 |
||
ソース: SDKI Analytics 専門家分析
自動液体ハンドラー市場成長要因
弊社の自動液体ハンドラー市場分析調査レポートによると、以下の市場動向と要因が市場成長の中核的な原動力として貢献すると予測されています。
-
研究室の労働力不足への対応
臨床検査室と研究室は、業務量の増加に伴い深刻な人手不足に直面しており、自動液体ハンドラーの導入は重要な解決策となっています。労働統計局は、検査技師と技術者の職種が2030年までに11%増加すると予測しています。
さらに、これらの機器は、試薬の分配やサンプルの転送などの反復作業を自動化することで、熟練したスタッフが監視、品質管理、複雑な分析に集中できるようにし、効率性とスループットを向上させることで、労働力の課題を市場の直接的な成長原動力にします。
-
ハイスループット研究の需要の高まり
創薬と診断におけるハイスループットスクリーニングと迅速なターンアラウンドの要求の高まりにより、自動液体ハンドラーへの投資が促進されています。NIH STRIDESイニシアチブの報告書によると、363以上の ペタバイト単位の生物医学研究データが 2,500 を超える研究プログラム間で共有されています。
さらに、このような大量のサンプルを扱うには、信頼性の高い自動サンプル処理が不可欠です。処理速度の向上、再現性の向上、そしてスケーラブルなワークフローの実現により、自動化されたサンプル処理は容易になっています。自動液体ハンドラーはこれらの要求に応えており、ラボが大量処理における効率性と精度を求める中で、市場を牽引しています。
サンプル納品物ショーケース
- 調査競合他社と業界リーダー
- 過去のデータに基づく予測
- 会社の収益シェアモデル
- 地域市場分析
- 市場傾向分析
レポートの洞察 - 自動液体ハンドラー市場の世界シェア
SDKI Analyticsの専門家によると、自動液体ハンドラー市場の世界シェアに関連するレポートの洞察は以下の通りです。
|
レポートの洞察 |
|
|
2026―2035年のCAGR |
7.2% |
|
2025年の市場価値 |
32.9億米ドル |
|
2035年の市場価値 |
72.5億米ドル |
|
過去のデータ共有 |
過去5年間 2024年まで |
|
将来予測 |
2035年までの今後10年間 |
|
ページ数 |
200+ページ |
ソース: SDKI Analytics 専門家分析
自動液体ハンドラー市場セグメンテーション分析
自動液体ハンドラー市場の展望に関連する様々なセグメントにおける需要と機会を説明する調査を実施しました。市場は技術別、アプリケーション別、モダリティ別、機器タイプ別、エンドユーザー別よってセグメント分割されています。
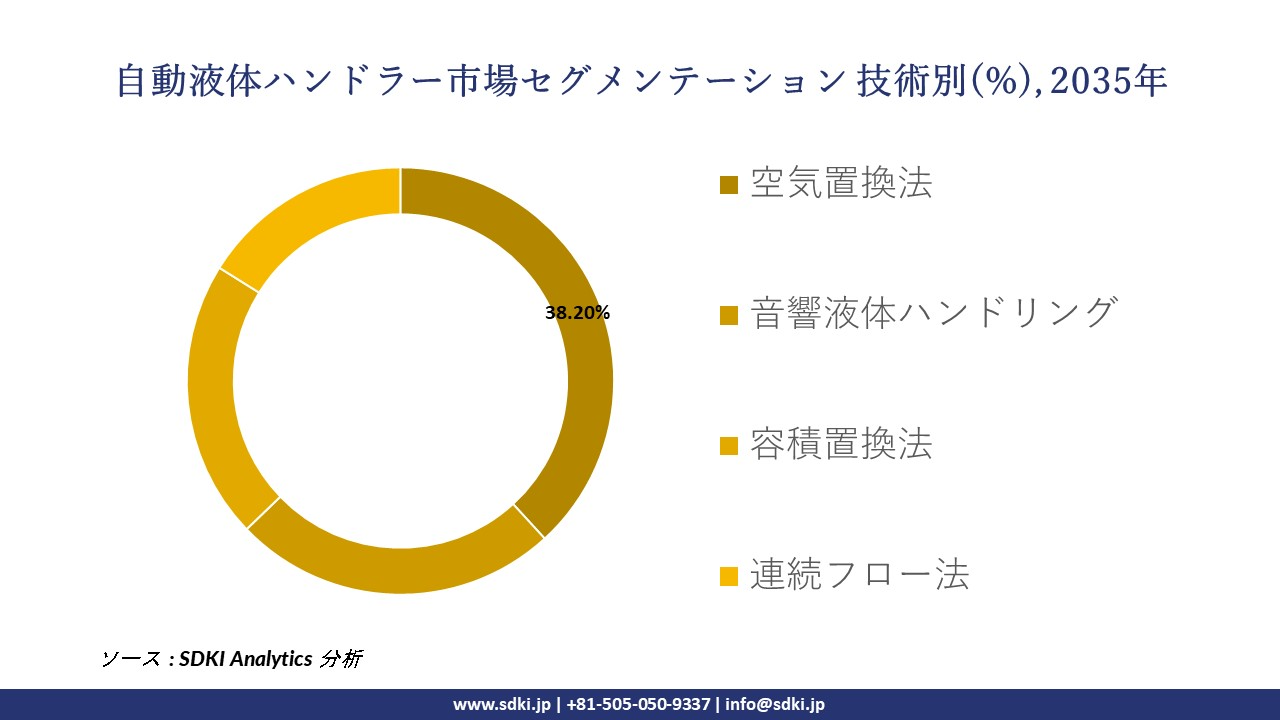
技術別ー
世界の自動液体ハンドラーの市場レポートによると、技術別に基づいて、空気置換法、音響液体ハンドリング、容積置換法、連続フロー法などに分割されています。
ここでは、空気置換法技術が予測期間中に市場を支配すると予想され、その高精度と汚染リスクの最小化により約 38.2% の市場シェアを占めると予想され、この技術は診断、創薬、ゲノミクス全体にわたる正確なサンプル浸透を可能にする上で重要な役割を果たします。
そのため、医薬品開発と商業化におけるシェア拡大を目指す新興バイオ医薬品企業の成長が急速に加速しており、これが自動液体ハンドラーへの技術アプリケーションを加速させていることが指摘されています。この主張を裏付ける証拠として、新興バイオ医薬品企業が創出する新規活性物質(NAS)の絶対数が、2019年の34件―2024年には41件に増加するというデータがあります。
アプリケーション別―
自動液体ハンドラー市場の調査展望によると、アプリケーション別に基づいて、創薬、ゲノミクス、臨床診断、法医学と環境検査といったサブセグメントに分割されています。その中でも、創薬分野は想定期間中に41.5%のシェアで市場を席巻すると見込まれています。
効率性と高度なスクリーニング機能、データ システムとロボット アームとの統合、試薬の消費量と廃棄物の発生量の削減といった要件により、創薬における自動液体ハンドラーのアプリケーションが盛んになります。
このセグメントの成長を牽引する要因としては、バイオ技術系スタートアップ企業の拡大、精密医療への投資増加、そして自動化アッセイプラットフォームの需要急増などが挙げられます。SDKI Analyticsの調査によると、自動化された液体ハンドラーはハイスループットラボにおいて重要であり、診断検査の効率を70%向上させ、人的ミスを55%削減し、ターンアラウンドタイムを大幅に短縮します。
以下は、自動液体ハンドラー市場に該当するセグメントのリストです。
|
親セグメント |
サブセグメント |
|
技術別 |
|
|
アプリケーション別 |
|
|
モダリティ別 |
|
|
機器タイプ別 |
|
|
エンドユーザー別 |
|
ソース: SDKI Analytics 専門家分析
世界の自動液体ハンドラー市場で調査された地域:
自動液体ハンドラー市場に関するこの調査レポートのために、以下の国と地域を調査しました。
|
地域 |
国 |
|
北米 |
|
|
ヨーロッパ |
|
|
アジア太平洋地域 |
|
|
ラテンアメリカ |
|
|
中東とアフリカ |
|
ソース: SDKI Analytics 専門家分析
自動液体ハンドラー市場の抑制要因
円滑な導入を阻む高額な資本と予算上の障壁は、市場の抑制要因の一つがあります。多くの研究室は、システムの初期費用の高さに加え、継続的なメンテナンスとソフトウェアのアップグレードの必要性から、自動液体ハンドラーの導入に躊躇しています。
この財政的負担は、研究資金が不安定な場合に特に深刻化します。例えば、2025年には、All of Usプログラムの2024年度予算は、助成金の打ち切りや資金削減により、2023年度から71%近く減少しました。その結果、自動化によって大幅な効率向上がもたらされるにもかかわらず、財政リスクと予算の制約が広範な導入を阻み、市場における大きな制約となっています。
自動液体ハンドラー市場 歴史的調査、将来の機会、成長傾向分析
自動液体ハンドラーメーカーの収益機会
自動液体ハンドラーに関連する収益機会の一部は次のとおりです。
|
機会エリア |
対象地域 |
成長の原動力 |
|
ゲノム研究の自動化 |
北米 |
強力な制度的資金と精密医療イニシアチブの優先順位付け |
|
バイオ医薬品製造の最適化 |
ヨーロッパ |
バイオ医薬品製造における再現性と自動化を求める規制の推進 |
|
臨床診断の拡大 |
アジア太平洋地域 |
人口密度の高い都市中心部における高スループット検査の需要の高まり |
|
学術研究インフラのアップグレード |
Mask |
|
|
開発業務受託機関(CRO)の成長 |
||
|
個別化医療パイプライン |
||
|
ワクチン開発の加速 |
||
|
環境試験自動化 |
||
ソース: SDKI Analytics 専門家分析
自動液体ハンドラーシェアの世界展開に向けた実現可能性モデル
弊社のアナリストは、自動液体ハンドラー市場の世界シェアを分析するために、世界中の業界専門家が信頼し、適用している有望な実現可能性モデルをいくつか提示しました。
|
実現可能性モデル |
地域 |
市場成熟度 |
医療システムの構造 |
経済発展段階 |
競争環境の密度 |
適用理由 |
|
学術パートナーシップモデル |
ラテンアメリカ |
新興 |
公共 |
現像 |
中 |
公立大学はバイオ技術教育の中心であり、技術提携に積極的です。 |
|
CRO統合モデル |
中東とアフリカ |
新生 |
ハイブリッド |
現像 |
低い |
地域の CRO は拡大しており、スケーラブルな自動化ソリューションを必要としています。 |
|
精密医療モデル |
北米 |
成熟した |
ハイブリッド |
発展した |
高い |
高度なヘルスケア システムは、パーソナライズされたケアにおける自動化の統合をサポートします。 |
|
バイオ医薬品コンプライアンスモデル |
Mask |
|||||
|
パンデミック対策モデル |
||||||
|
環境モニタリングモデル |
||||||
|
大学インキュベーターモデル |
||||||
|
高スループット診断モデル |
||||||
ソース: SDKI Analytics 専門家分析
市場傾向分析と将来予測:地域市場の見通しの概要
➤ 北米の自動液体ハンドラー市場規模:
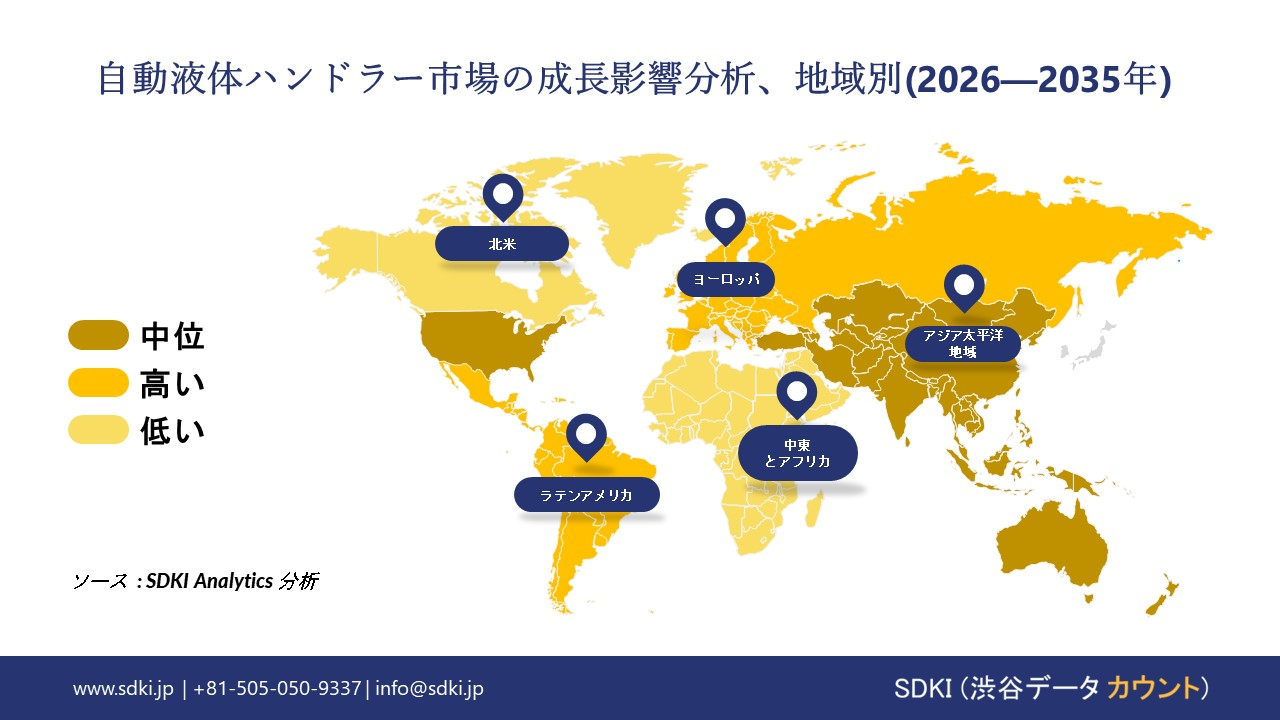
自動液体ハンドラー市場 北 アメリカは予測期間中、約38.0%という最大の市場シェアを占めると予想されています。この力強い成長は、成熟したライフ サイエンスセクター、バイオ医薬品への堅調な投資、そしてハイスループットスクリーニングと自動化に注力するインフラ整備によって支えられています。
米国連邦政府の研究機関は引き続き多額の資金提供を行っており、例えば国立衛生研究所(NIH)への資金提供は928.9億米ドルの経済活動を生み出しており、これは基盤となる研究インフラの規模を反映しています。さらに、この地域の規制環境は、トレーサビリティと再現性を確保するための自動化を奨励しており、ALH技術の需要を高めています。
- 北米の自動液体ハンドラー市場の市場強度分析:
自動液体ハンドラー市場に関連する国の市場強度分析は次のとおりです。
|
米国 |
カナダ |
|
|
市場の成長可能性 |
強い |
適度 |
|
規制環境の複雑さ |
複雑な |
標準 |
|
価格体系 |
市場主導型 |
ハイブリッド |
|
熟練した人材の可用性 |
Mask |
|
|
標準及び認証フレームワーク |
||
|
イノベーションエコシステム |
||
|
技術統合率 |
||
|
市場参入障壁 |
||
|
投資環境 |
||
|
サプライチェーン統合 |
||
|
競争の激しさ |
||
|
顧客基盤の高度化 |
||
|
インフラの準備 |
||
|
貿易政策の影響 |
||
ソース: SDKI Analytics 専門家分析
➤ ヨーロッパの自動液体ハンドラー市場規模:
自動液体ハンドラー市場 ヨーロッパは予測期間中に急速な成長を遂げると予測されています。ヨーロッパ地域は、医薬品と診断分野の力強い発展、ライフ サイエンス研究への政府の強力な資金提供、そしてEUの規制枠組み(EU域内における規制の枠組みなど)によって成長しています。 自動化とトレーサビリティの向上を推進する IVDR など、さまざまな技術が開発されています。
OECによれば、2023年にはドイツはヨーロッパで実験用ガラス器具の主要輸出国となり、輸出額は262百万米ドルに達します。 百万ドル規模の堅牢な研究インフラは、研究及び診断ワークフローの効率向上を目的とした自動液体ハンドラーの導入拡大を支えています。さらに、 EU域内の国境を越えた連携と標準化により、自動サンプル処理ワークフローの導入が促進されています。
- ヨーロッパ自動液体ハンドラー市場の市場強度分析:
自動液体ハンドラー市場に関連する国の市場強度分析は次のとおりです。
|
カテゴリ |
イギリス |
ドイツ |
フランス |
|
市場の成長可能性 |
適度 |
強い |
強い |
|
規制環境の複雑さ |
複雑な |
標準 |
標準 |
|
インセンティブと補助金の支援 |
高い |
適度 |
高い |
|
熟練した人材の可用性 |
Mask |
||
|
研究開発とイノベーションのエコシステム |
|||
|
EV普及率 |
|||
|
サプライチェーンのレジリエンス |
|||
|
製造基盤の強さ |
|||
|
技術統合率 |
|||
|
市場参入障壁 |
|||
|
投資環境 |
|||
|
競争の激しさ |
|||
|
アフターマーケットの成熟度 |
|||
|
貿易政策の影響 |
|||
ソース: SDKI Analytics 専門家分析
➤ アジア太平洋地域の自動液体ハンドラー市場規模:
自動液体ハンドラー市場 アジア 太平洋地域は、予測期間中に7.0%のCAGRで成長し、最も急速な市場となることが予想されています。この地域の成長は、中国、インド、日本、南アフリカなどの主要国における製薬業界やバイオ技術業界の急成長などによって牽引されています。 韓国では、医療インフラへの投資が増加し、診断ラボの数も増加しています。
人口の多さ、診断検査数の増加、そして人的ミスの削減の必要性から、この地域では自動液体処理ソリューションの需要が高まっています。さらに、地域政府の取り組みはラボの自動化に重点を置いており、導入拡大を後押ししています。例えば、インドの診断業界は今後5年間で複利年間成長率(CAGR)14%の成長が見込まれており、自動化ラボ技術への投資拡大を後押ししています。
- アジア太平洋地域の自動液体ハンドラー市場の市場強度分析:
自動液体ハンドラー市場に関連する国の市場強度分析は次のとおりです。
|
カテゴリ |
日本 |
韓国 |
マレーシア |
中国 |
インド |
|
市場の成長可能性 |
中程度→ 強い |
強い |
適度 |
強い |
中程度→ 強い |
|
EV普及の勢い |
中くらい |
高い |
低い→ 中くらい |
高い |
中くらい |
|
ローカルコンテンツ要件 |
適度 |
適度 |
適度 |
厳格→ 適度 |
中程度→ 厳しい |
|
半導体リスクの影響 |
Mask |
||||
|
OEM生産基盤の強さ |
|||||
|
アフターマーケットの成熟度 |
|||||
|
製造コスト競争力 |
|||||
|
研究開発とイノベーションのエコシステム |
|||||
|
サプライチェーン統合 |
|||||
|
政策とインセンティブのサポート |
|||||
|
市場参入障壁 |
|||||
|
輸出志向 |
|||||
|
貿易政策の影響 |
|||||
ソース: SDKI Analytics 専門家分析
自動液体ハンドラー業界概要と競争ランドスケープ
自動液体ハンドラー市場のメーカーシェアを支配する世界トップ 10 社は次のとおりです。
|
会社名 |
本社所在地国 |
自動液体ハンドラーとの関係 |
|
Thermo Fisher Scientific |
米国 |
研究室向けの自動液体処理システムを提供 |
|
Hamilton Company |
米国 |
ロボット液体処理ワークステーションを製造 |
|
Agilent Technologies |
米国 |
自動液体処理機器を提供する |
|
Aurora Biomed |
Mask |
|
|
Eppendorf |
||
|
Tecan Group |
||
|
BioTek Instruments |
||
|
PerkinElmer |
||
|
SPT Labtech |
||
|
HiMedia Laboratories |
||
ソース: SDKI Analytics 専門家分析及び企業ウェブサイト
自動液体ハンドラーの世界及び日本の消費者上位 10 社は次のとおりです。
| 主要消費者 | 消費単位(数量) | 製品への支出 – 米ドル価値 | 調達に割り当てられた収益の割合 |
|---|---|---|---|
| Thermo Fisher Scientific |
|
||
| JEOL Ltd. | |||
| XXXX | |||
| XXXXX | |||
| xxxxxx | |||
| xxxxxxxx | |||
| xxxxx | |||
| xxxxxxxx | |||
| xxxxxx | |||
| XXXXX | |||
日本の自動液体ハンドラー市場のメーカーシェアを占める上位10社は次のとおりです。
|
会社名 |
事業状況 |
自動液体ハンドラーとの関係 |
|
Shimadzu Corporation |
日本原産 |
自動化されたサンプル調製及び液体処理システムを提供します |
|
Hitachi High-Tech |
日本原産 |
液体処理を含むラボの自動化を提供 |
|
JEOL Ltd. |
日本原産 |
自動サンプル処理システムを提供 |
|
Wako Pure Chemical Industries (FUJIFILM) |
Mask |
|
|
Nacalai Tesque |
||
|
Yamato Scientific |
||
|
Kanto Kagaku |
||
|
Advantec MFS |
||
|
Tokyo Rikakikai (EYELA) |
||
|
AS ONE Corporation |
||
ソース: SDKI Analytics 専門家分析及び企業ウェブサイト
自動液体ハンドラー 市場 包括的企業分析フレームワーク
市場内の各競合他社について、次の主要領域が分析されます 自動液体ハンドラー 市場:
- 会社概要
- リスク分析
- 事業戦略
- 最近の動向
- 主要製品ラインナップ
- 地域展開
- 財務実績
- SWOT分析
- 主要業績指標
自動液体ハンドラー市場最近の開発
世界及び日本における自動液体ハンドラー市場に関連する最近の商業的発売及び技術の進歩の一部は次のとおりです。
|
会社名 |
発売の詳細 |
|
Agilent Technologies |
2025年1月、ABB Roboticsと分析ラボ技術ソフトウェアの世界的リーダーであるAgilent Technologiesは、自動化ラボソリューションの提供に関する協業契約を締結しました;これにより、両社の技術のメリットを融合し、製薬、バイオ技術、エネルギー、食品と飲料など、複数の業界の企業が研究や品質管理などのプロセスをより迅速かつ効率的にすることで、ラボ業務を変革できるようになります。 |
|
Shimadzu Corporation |
2025年9月、組織はiシリーズラインの新モデル(LC-2070及びLC-2080)を導入しました。起動時の自動システム診断と、自動化されたカラム情報管理のためのインテリジェントなカラム管理プラットフォーム( iCMP )を備えており、運用効率と信頼性が向上しています。 |
ソース:各社プレスリリース
目次

関連レポート
よくある質問

- 2020ー2024年
- 2026-2035年
- 必要に応じて日本語レポートが入手可能





