ジカウイルス(ZIKV)感染は、アフリカと東南アジアで風土病であることが知られている

ジカウイルス(ZIKV)感染は、アフリカと東南アジアで風土病であることが知られている。その結果、太平洋地域全体に広がったウイルスは、世界中の先進国と新興国での発生につながった。世界保健機関(WHO)によると、約84の国/地域がZIKV感染に感染しているか、または以前に感染しています。アジア太平洋地域(特にインド)と南米諸国でジカウイルス感染の大規模な発生がありました。疾病管理予防センターによると、メキシコ、南米、中央、サハラ以南のアフリカ諸国、東南アジアのいくつかの国は、ジカウイルス感染の増殖の危険にさらされている主要国です。したがって、ジカウイルス感染の有病率の上昇は、予測期間中に調査された市場の成長を促進する主な要因の1つです
調査された市場の他の顕著な推進力は、バイオ製薬会社によるR&Dの増加と、診断テストにおける技術的進歩です
主な市場動向
血清学的検査セグメントは、予測年
でより良い成長を示すと予想されます
ジカウイルス検査市場は、検査の種類とエンドユーザーに基づいてセグメント化されています。テストタイプは、分子テストと血清学的テストにさらにセグメント化されます。血清学的検査は、急性および後期段階のジカウイルス感染を検出できるため、嗜好が高まるため、増加すると予想されます。さらに、新しい迅速な検出血清学的診断は、より短いターンアラウンドタイムと、他のフラビウイルスに対するジカの鑑別診断を提供する。さらに、妊娠初期のジカは先天性欠損症のリスクを高めます。CDCによると、先天性欠損症の症例は、妊娠の最初の三半期に母親がジカウイルスに感染した赤ちゃんの割合が高くなりました。このウイルスは、感染した妊婦から胎児に伝染する可能性があり、小頭症やその他の重度の脳異常を引き起こします。これにより、妊婦のジカウイルス検査が促され、市場の成長がさらに加速しました
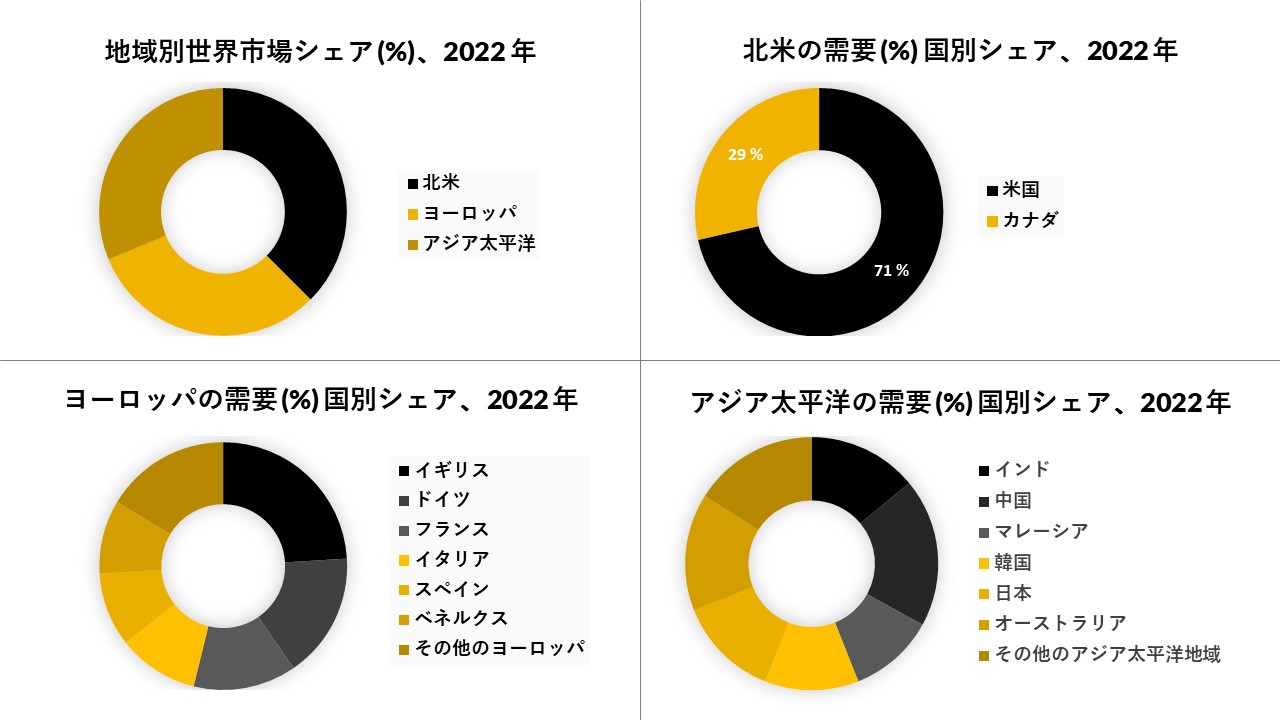
北米は市場を支配しており、予測期間中に同じ傾向に従うと予想されています
北米は、調査した市場を支配すると予想されます。この成長は、テストキットの利点に対する意識の高まりに起因する可能性があります。疾病管理予防センター(CDC)、パンアメリカン保健機関(PAHO)などの政府機関は、主にジカウイルス感染の予防に関与してきました。CDCによると、米国の州では妊娠中にジカウイルス感染が確認された女性の赤ちゃんの約20人に2人(10%)、米国の領土では約20人に1人(5%)がジカ熱関連の先天性欠損症を患っていました。CDCは、いくつかの地域でジカ熱の流行と戦うための資金を受けています。さらに、発達した医療インフラと、ジカウイルスの検査におけるいくつかのバイオ製薬会社による急速な技術進歩は、市場にプラスの影響を与えると予想されています
競争環境
ジカウイルス検査市場のグローバルプレーヤーは、アボットラボラトリーズ、アルトナダイアグノスティックスGmbH、ケムバイオ診断システムズ、ELITechGroup、F.ホフマン・ラ・ロシュ、ジェネカム、ルミネックス・コーポレーション、ノバシット・グループ、クエスト・ダイアグノスティックス、シーメンスAG.
このレポートを購入する理由:
- エクセル形式の市場予測(ME)シート
- クライアントの要件に従ってカスタマイズを報告
- 3ヶ月のアナリストサポート

北米(米国およびカナダ)、ラテンアメリカ(ブラジル、メキシコ、アルゼンチン、その他のラテンアメリカ)、ヨーロッパ(英国、ドイツ、フランス、イタリア、スペイン、ハンガリー、ベルギー、オランダおよびルクセンブルグ、NORDIC(フィンランド、スウェーデン、ノルウェー) 、デンマーク)、アイルランド、スイス、オーストリア、ポーランド、トルコ、ロシア、その他のヨーロッパ)、ポーランド、トルコ、ロシア、その他のヨーロッパ)、アジア太平洋(中国、インド、日本、韓国、シンガポール、インドネシア、マレーシア) 、オーストラリア、ニュージーランド、その他のアジア太平洋地域)、中東およびアフリカ(イスラエル、GCC(サウジアラビア、UAE、バーレーン、クウェート、カタール、オマーン)、北アフリカ、南アフリカ、その他の中東およびアフリカ)