クリティカルケア診断市場は、主に慢性および生活習慣病の世界的な有病率の増加と診断技術の進歩によって牽引されています

クリティカルケア診断市場は、主に慢性および生活習慣病の世界的な有病率の増加と診断技術の進歩によって牽引されています。保健図書館によると、米国では約8400万人が何らかの形の心血管疾患に罹患しており、1日に約2200人が死亡し、平均40秒ごとに1人が死亡していると推定されています
さらに、早期診断と治療に対する人々の意識の高まり、電子医療記録(EHR)との相互運用性によるデータ管理と接続性の使用の増加、遠隔医療サービスとリモート通信の採用の増加は、クリティカルケア診断市場の成長を促進している他の要因です。しかし、診断テストの高コストとクリティカルケア診断を扱う熟練した専門家の欠如は、市場の成長を妨げる可能性があります
主な市場動向
緊急治療室は市場をリードすることが期待されています
重症患者が推奨される緊急治療室に対する世間の需要は、着実に増加しています。救急科における重症患者の生存率は、疾患の早期発見と治療の進歩に直接関係しています。メリーランド大学がScience Daily誌に発表した2017年の最近の記事によると、救急部門が提供する救命救急医療の割合が大幅に増加し、米国の全医療のほぼ半分が救急部門によって提供されました.
米国保健福祉省2018によると、2017年には、米国の成人の約18.6%が過去12ヶ月間に1回以上の救急部門の訪問を受けました。さらに、欧州諸国でも同様の緊急治療室数の増加パターンが観察できますが、インド、中国、日本などの新興経済国ではその数が大きく異なります
さらに、高齢化による事故や慢性疾患による怪我の負担の増大は、予測期間中のクリティカルケア診断市場の成長を促進すると予測されています
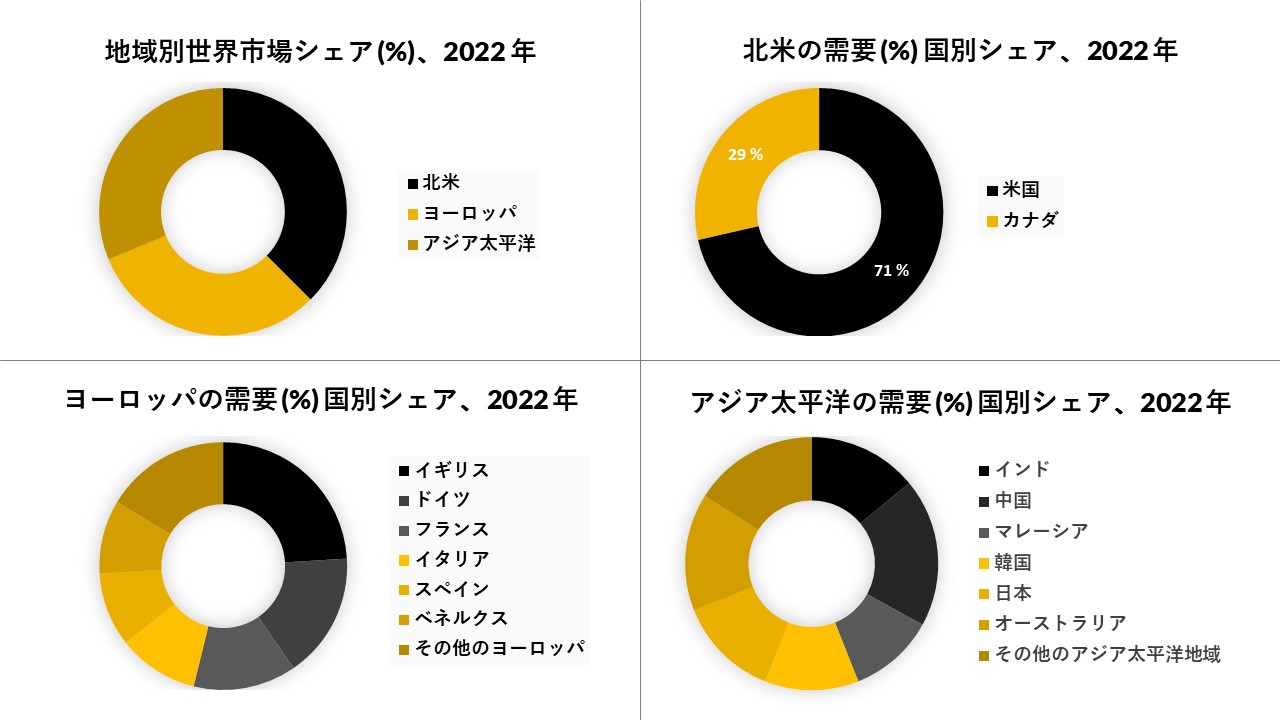
北米は予測期間を支配すると予想されています
北米は安定した成長を遂げていますが、地域によって大きなばらつきがあります。米国の病院は、手術室の物理的インフラに多額の投資を行ってきました。米国の病院から生み出される収益の60%以上は、平均的な病院の手術室での活動から来ています.
この技術のアップグレードの必要性は、クリティカルケアの診断と管理にとって依然として不可欠です。この要因は、手術室の手順を含む長期入院の高コストを伴います。病院の費用自体は非常に高いです。高い医療費、より良いインフラ、政府による追加の関与は、北米の市場の成長を促進すると予想されています
競争環境
市場プレーヤーは、クリティカルケア診断市場が大幅な成長を獲得するのに役立つ資金調達とコラボレーションを通じて、政府の開始に関与しています。トッププレーヤーはまた、新製品の発売、パートナーシップ、コラボレーションなどに従事し、市場での存在感を高めています。例えば、2018年1月、GEヘルスケアはロシュ社と戦略的で長期的なパートナーシップを結び、がん患者および救命救急患者向けのデジタル臨床意思決定支援ソリューションを共同開発しました。市場の主要プレーヤーは、アボット、ベックマン・コールター、ロシュなどです
このレポートを購入する理由:
- エクセル形式の市場予測(ME)シート
- 3ヶ月のアナリストサポート

北米(米国およびカナダ)、ラテンアメリカ(ブラジル、メキシコ、アルゼンチン、その他のラテンアメリカ)、ヨーロッパ(英国、ドイツ、フランス、イタリア、スペイン、ハンガリー、ベルギー、オランダおよびルクセンブルグ、NORDIC(フィンランド、スウェーデン、ノルウェー) 、デンマーク)、アイルランド、スイス、オーストリア、ポーランド、トルコ、ロシア、その他のヨーロッパ)、ポーランド、トルコ、ロシア、その他のヨーロッパ)、アジア太平洋(中国、インド、日本、韓国、シンガポール、インドネシア、マレーシア) 、オーストラリア、ニュージーランド、その他のアジア太平洋地域)、中東およびアフリカ(イスラエル、GCC(サウジアラビア、UAE、バーレーン、クウェート、カタール、オマーン)、北アフリカ、南アフリカ、その他の中東およびアフリカ)