オーファンドラッグ市場は、予測期間中に11.5%のCAGRを登録すると予想されています

オーファンドラッグ市場は、予測期間中に11.5%のCAGRを登録すると予想されています。この市場の成長を担う要因には、オーファンドラッグ開発者の市場独占性、希少疾患の罹患率の上昇、および有利な政府の政策が含まれます
・近年、世界人口における希少疾患の罹患率は増加の一途をたどっています。この問題に取り組むために、開発途上国と先進国の両方が希少疾患のための薬の開発を促進するだけでなく、これらの薬が患者にとって容易に利用可能であることを確認するための規制を策定しました。現在、非オーファンドラッグの平均承認期間は約13ヶ月で、オーファンドラッグについても約10ヶ月ではるかに短くなっています
欧州連合(EU)はこれらの薬物の開発について同様の政策を実施しており、欧州各国政府はこれらの政策を自国で個別に実施している。これらのいくつかは、イタリアのAIFA5%基金、ベルギーの特別連帯基金、およびフランスの一時的な使用許可です。アジア太平洋地域の国々でさえ、日本、韓国、台湾などの国々と同様のステップを踏んでおり、改訂されたオーファンドラッグ規則やオーファンドラッグガイドライン(2003)などのイニシアチブを実施しています。これらのイニシアチブは、患者と製薬会社の両方に税額控除と補助金を提供しました。世界中のさまざまな国におけるこれらの政策は、近年健全な速度で成長している市場を進化させてきました
主な市場動向
バイオオーファンドラッグは、最高の市場シェアを占めると予想されています
薬物セグメンテーションの種類の下では、生物学的オーファンドラッグが最も高い市場シェアを記録する可能性があり、このセグメントは予測期間中に良好なペースで成長すると予想されます。この高い市場シェアは、非生物学的製剤と比較して生物学的薬物のコストが高いためです。複数の適応症に対する生物学的オーファンドラッグの承認の最近の傾向は、成長をもたらし、新規および確立された市場プレーヤーがこの市場に参入する有望な要因となっています。もう一つの理由は、生物学的薬物が希少疾患の治療に長い間使用されてきたことです。最も焦点を絞った孤児疾患、すなわち先進国で高い罹患率を有する癌は、副作用の少ない生物学的薬物によって効果的に治療されてきた
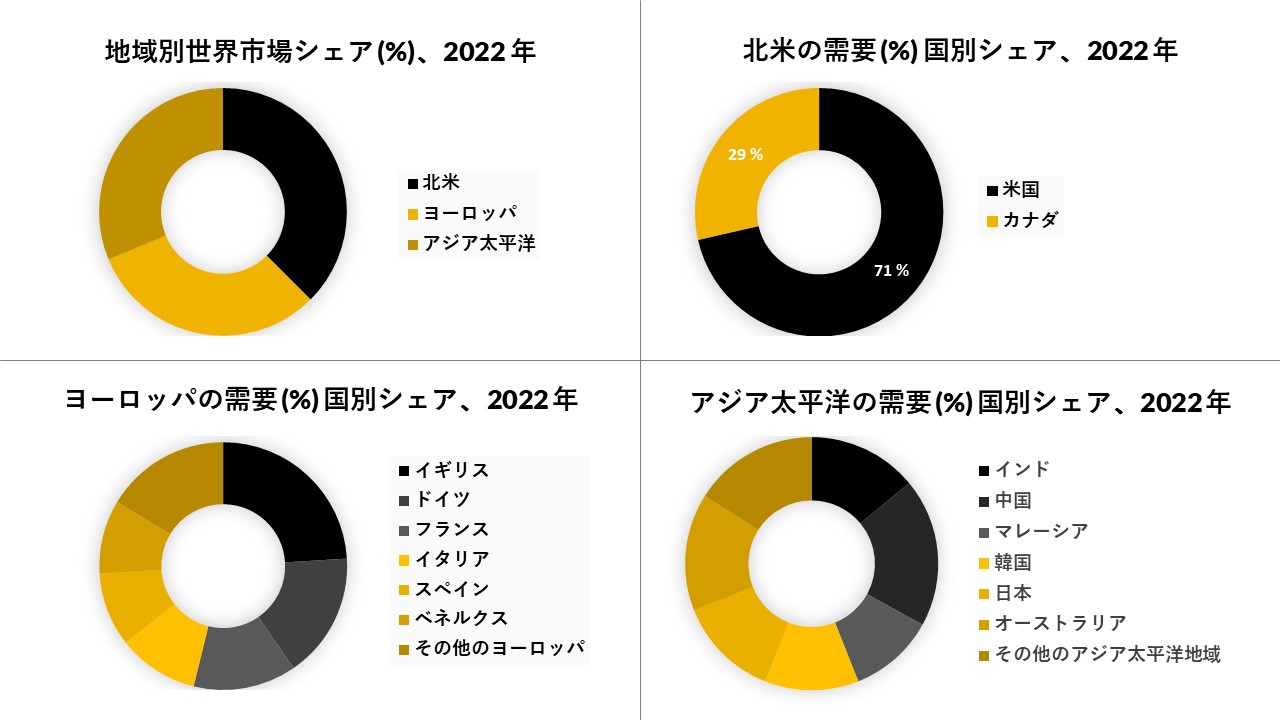
北米が市場を支配し、予測期間
で同じことをすると予想されています
北米は現在、オーファンドラッグの市場を支配しており、今後数年間は拠点を継続すると予想されています。北米地域では、米国が最大の市場シェアを占めています。市場成長の理由の1つは、米国では、医薬品指定オーファンドラッグステータスが、特定の適応症、税額控除、および使用料免除に対するFDAの承認を受けて、7年間の販売独占権を受け取ることです。2013年以来、新しい適応症の承認が大幅に増加しました.
競争環境
オーファンドラッグ市場は適度に競争が激しく、かなりの数の主要プレーヤーで構成されています。市場シェアの面では、現在、いくつかの主要プレーヤーが市場を支配しています。毎年病気の数と症例数が増加しているため、いくつかの小規模なプレーヤーが市場に参入し、かなりのシェアを保持しています。市場の主要プレーヤーには、Teva Pharmaceutical Industries Ltd.、Celgene、Merck & Co. Inc.、Bristol-Myers Squibb Company、F. Hoffmann-La Roche AG><などがあります。
このレポートを購入する理由:
- エクセル形式の市場予測(ME)シート
- 3ヶ月のアナリストサポート

北米(米国およびカナダ)、ラテンアメリカ(ブラジル、メキシコ、アルゼンチン、その他のラテンアメリカ)、ヨーロッパ(英国、ドイツ、フランス、イタリア、スペイン、ハンガリー、ベルギー、オランダおよびルクセンブルグ、NORDIC(フィンランド、スウェーデン、ノルウェー) 、デンマーク)、アイルランド、スイス、オーストリア、ポーランド、トルコ、ロシア、その他のヨーロッパ)、ポーランド、トルコ、ロシア、その他のヨーロッパ)、アジア太平洋(中国、インド、日本、韓国、シンガポール、インドネシア、マレーシア) 、オーストラリア、ニュージーランド、その他のアジア太平洋地域)、中東およびアフリカ(イスラエル、GCC(サウジアラビア、UAE、バーレーン、クウェート、カタール、オマーン)、北アフリカ、南アフリカ、その他の中東およびアフリカ)