世界のインフルエンザ診断市場は、インフルエンザおよび関連する慢性疾患の罹患率の増加、インフルエンザ診断のための研究資金の増加、インフルエンザの早期診断および制御に対する需要の高まりなどにより、6.90%のCAGRを目撃しています.
インフルエンザは、ヒト集団における罹患率および死亡率の重要な原因として認識されており、その結果生じる健康および経済的影響を軽減することを目的とした診断法の開発および使用につながっている

世界のインフルエンザ診断市場は、インフルエンザおよび関連する慢性疾患の罹患率の増加、インフルエンザ診断のための研究資金の増加、インフルエンザの早期診断および制御に対する需要の高まりなどにより、6.90%のCAGRを目撃しています.
インフルエンザは、ヒト集団における罹患率および死亡率の重要な原因として認識されており、その結果生じる健康および経済的影響を軽減することを目的とした診断法の開発および使用につながっている。WHOによると、2018年にインフルエンザは、主に高リスクの患者に重度の病気や死亡を引き起こし、軽度から重症、さらには死亡までの範囲でした。また、この年間増加は、世界中で約300万〜500万例の重篤な疾患症例、および約290000〜650000人の死亡をもたらすと予測されています
さまざまなイニシアチブと研究資金が市場の成長を支えています。例えば、国立アレルギー感染症研究所(NIAID)は、季節性インフルエンザおよびパンデミックインフルエンザに対するワクチン、診断薬、および治療法の開発を知らせる研究を含む、インフルエンザ研究の幅広いポートフォリオを支援してきました。同様に、2018年には、ヒトワクチンプロジェクトと呼ばれる、学術、政府、業界の医学研究者間の非営利団体が、ユニバーサルインフルエンザワクチンイニシアチブを立ち上げました。したがって、インフルエンザ診断のための政府や企業による研究資金の増加が市場を牽引しています
しかし、すべての迅速な抗原インフルエンザ検査が承認のための新しいFDA要件を満たしていることを確認することが重要です。したがって、厳しいFDA規制は、市場にとって重要な制約として機能します
主な市場動向
インフルエンザ迅速診断検査(RIDT)は、検査タイプセグメント
で最高の市場シェアを保持すると予想されています
インフルエンザ診断市場の検査タイプセグメントでは、すべての従来の診断検査の中で、迅速なインフルエンザ診断検査(RIDT)が最大の市場規模を持つと考えられており、予測期間中に6.87%のCAGRを目撃すると予想されています
インフルエンザ迅速診断検査(RITT)は免疫アッセイであり、呼吸器検体中のインフルエンザAおよびBウイルス核タンパク質抗原の存在を同定し、定性的に結果を提供するのに役立ちます。これは、患者を診断することができます インフルエンザ様疾患で存在しています それは症状の発症の最初の4日以内に行われる場合 主に.
RIDTの人気が高まっているのは、より速い結果と簡単な使用の利点によるものです。一部のRIDTは、オフィス/ベッドサイドでの使用も承認されています。RIDTセグメントはまた、ポイントオブケア環境での一般的な使用の結果として、急速な成長を目撃することが期待されています.
しかし、RIDTに関連する主な欠点の1つは、感度が低いから中程度(50〜70%)であるため、偽陰性の結果を与えることです。医療政策立案者は、新型H1N1インフルエンザの流行が発生した2009年に、その感受性の低さの影響を認識し始めました.
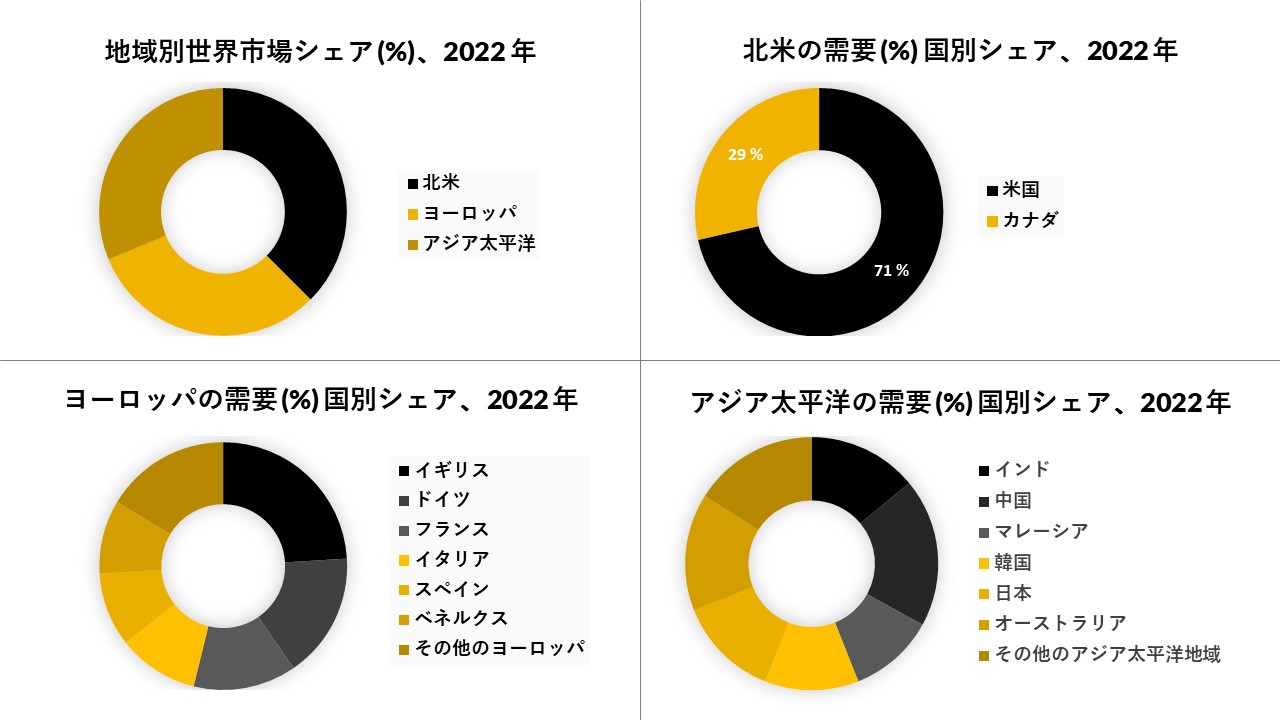
北米は市場を支配しており、予測期間
北米は現在、インフルエンザ診断薬の市場を支配しており、今後数年間は拠点を継続すると予想されています
疾病管理予防センター(CDC)が公開したデータによると、RIDTは地域全体で一般的に入手可能です。テストの可用性が広い理由は、結果を生成するために必要な期間が短縮されたことです。RIDTは15分で結果を得ることができます。インフルエンザ関連の入院と小児死亡の増加と、この疾患をよりよく管理したいという政府機関の関心により、北米のインフルエンザ診断市場は将来的に成長すると予想されています
競争環境
インフルエンザ診断薬市場は非常に競争が激しく、いくつかの主要プレーヤーで構成されています。市場シェアの面では、現在市場を支配している主要プレーヤーはほとんどありません。Coris BioConcept、DiaSorin SpA、F. Hoffmann-La Roche AG、Luminex Corporation、Meridian Bioscience Inc.、Quidel Corporation、Sekisui Diagnosticsなどの企業が市場で大きなシェアを占めています
最近、市場ではさまざまな開発が行われています。例えば、積水ダイアグノスティックスは2018年3月、メサバイオテックと共同で開発したシラリスインフルエンザAおよびB検査を開始しました。また、2017年10月、アボット・ラボラトリーズは診断装置およびサービスプロバイダーのAlere Inc.(Alere)を買収し、同社の診断プレゼンスを強化しました
このレポートを購入する理由:
- エクセル形式の市場予測(ME)シート
- 3ヶ月のアナリストサポート

北米(米国およびカナダ)、ラテンアメリカ(ブラジル、メキシコ、アルゼンチン、その他のラテンアメリカ)、ヨーロッパ(英国、ドイツ、フランス、イタリア、スペイン、ハンガリー、ベルギー、オランダおよびルクセンブルグ、NORDIC(フィンランド、スウェーデン、ノルウェー) 、デンマーク)、アイルランド、スイス、オーストリア、ポーランド、トルコ、ロシア、その他のヨーロッパ)、ポーランド、トルコ、ロシア、その他のヨーロッパ)、アジア太平洋(中国、インド、日本、韓国、シンガポール、インドネシア、マレーシア) 、オーストラリア、ニュージーランド、その他のアジア太平洋地域)、中東およびアフリカ(イスラエル、GCC(サウジアラビア、UAE、バーレーン、クウェート、カタール、オマーン)、北アフリカ、南アフリカ、その他の中東およびアフリカ)