焦点分節性糸球体硬化症(FSGS)治療市場は、主に疾患負担の増加、希少疾患への関心の高まり、およびR&D活動の増加によって牽引されています

焦点分節性糸球体硬化症(FSGS)治療市場は、主に疾患負担の増加、希少疾患への関心の高まり、およびR&D活動の増加によって牽引されています。他の糸球体疾患と比較して、FSGSの有病率は世界中で急速に増加していることがわかっています
NephCureによると、毎年、FSGSと診断されている約5400人以上の患者がおり、約20000人のESRD患者がFSGSのために早期に罹患していました。さらに、アフリカ系の人々は5倍の診断率を有し、ステロイドに反応しない人々は腎臓移植または透析のために移動し、患者の30%〜40%がFSGS疾患を再発する。さらに、カリフォルニア大学によると、ESRDは毎年5%増加しています。これにより、ドナーの腎臓の必要性は毎年8%増加しています
先進国と新興市場の両方で、腎臓移植の需要が高まり、腎臓の機能不全による入院率が高まっています。これは、腎臓病の治療中にFSGS疾患の初期段階を診断する需要を増加させる。したがって、糸球体関連疾患の負担は、予測期間中のFSGS市場の需要を牽引しています
主な市場動向
原発性FSGSは、疾患型セグメント
において高いCAGRを記録すると予想されます。
原発性FSGSは、成人における特発性糸球体疾患の最も一般的な原因の1つとなっている。原発性FSGSは、通常、「特発性」であり、既知の、明確な、または明白な理由なしに疾患が上昇することを意味する。それはいくつかの原型的特徴を有し、青年および若年成人において最も一般的な形態である。一次FSGSに対する現在の治療法には、グルココルチコイドおよびカルシニューリン阻害剤などの免疫抑制剤が含まれる。ステロイド療法は、経口プレドニゾンによる最も基本的かつ標準的な治療法の一つです。パイプラインに潜在的な医薬品候補を持つ主要な新興企業のいくつかは、Chemocentryx、Reata、Pharmaceuticals、Retrophin Inc.、Vertex Pharmaceuticals.
これは希少疾患であるため、専門の臨床段階の企業はより多くの資金を得る可能性が高く、戦略的パートナーシップの機会を活用しています。さらに、両社は、従来の薬物および外科的治療に対する解決策として、より標的を絞った治療法に研究開発努力を向けています。したがって、上記の声明により、セグメントは予測期間中に急速な成長を遂げることが観察されています.
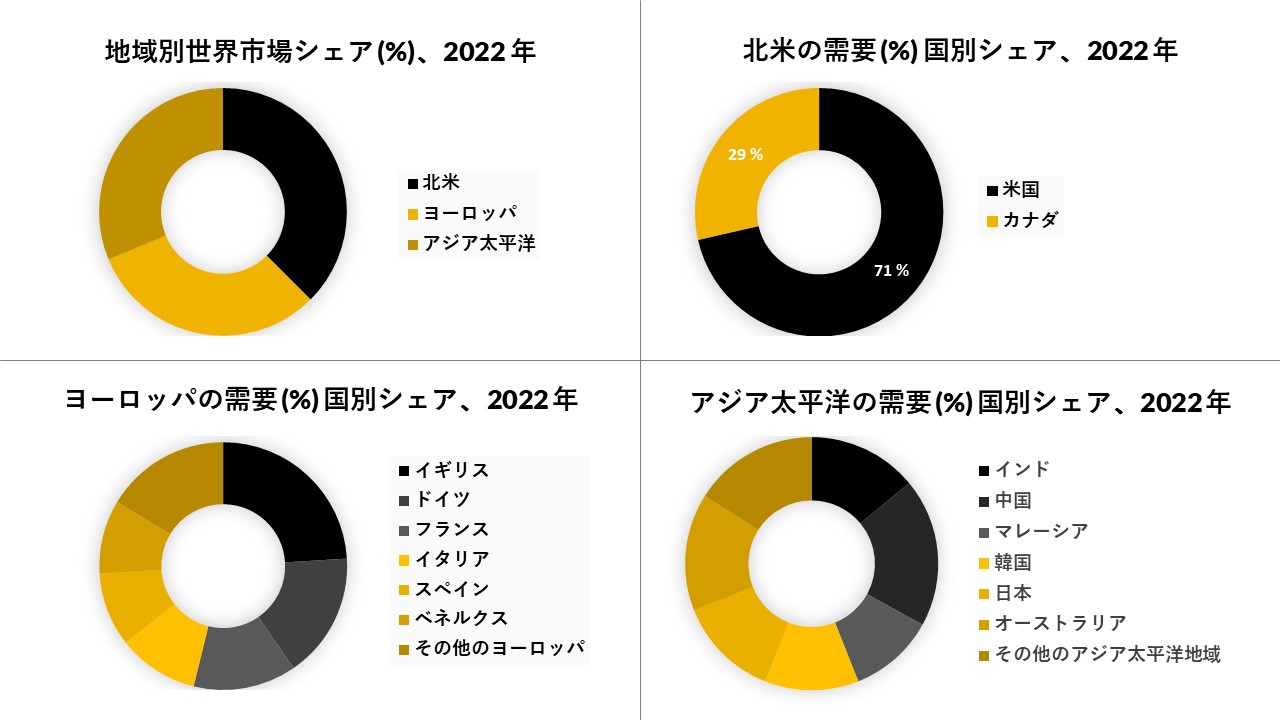
北米はFSGS治療市場で最大のシェアを持つと予想されています
北米は、適切に構造化された医療業界の存在と、より広いカバレッジを備えた償還ポリシーの可用性のために、市場で主導的なシェアを占めています。FSGSの普及率の高まり、最先端の医療インフラの存在、新しい医療技術の高い採用率、および償還の存在が市場の成長を促進しています
さらに、米国は、いくつかの製造業者が臨床試験を実施するための最初の選択肢の1つです。FSGS臨床試験の大部分は米国で実施済みまたは進行中であり、試験中の薬物が支配的であった試験の資金提供に大学や研究センターが関与しています。FSGSについては、いくつかの臨床試験と医薬品の臨床試験の承認が進行中であり、予測期間中に高い成長を示しています
競争環境
FSGS治療市場は適度に統合されており、透析やその他の診断に関連する製品を提供する医療機器事業に参入しているグローバルプレーヤーで構成されています。この市場に関与する主要な製薬会社は、自社製品をパイプラインに投入しており、今後数年間で製品をリリースすることが期待されています
このレポートを購入する理由:
- エクセル形式の市場予測(ME)シート
- 3ヶ月のアナリストサポート

北米(米国およびカナダ)、ラテンアメリカ(ブラジル、メキシコ、アルゼンチン、その他のラテンアメリカ)、ヨーロッパ(英国、ドイツ、フランス、イタリア、スペイン、ハンガリー、ベルギー、オランダおよびルクセンブルグ、NORDIC(フィンランド、スウェーデン、ノルウェー) 、デンマーク)、アイルランド、スイス、オーストリア、ポーランド、トルコ、ロシア、その他のヨーロッパ)、ポーランド、トルコ、ロシア、その他のヨーロッパ)、アジア太平洋(中国、インド、日本、韓国、シンガポール、インドネシア、マレーシア) 、オーストラリア、ニュージーランド、その他のアジア太平洋地域)、中東およびアフリカ(イスラエル、GCC(サウジアラビア、UAE、バーレーン、クウェート、カタール、オマーン)、北アフリカ、南アフリカ、その他の中東およびアフリカ)