調査対象のバイオ分析検査サービス市場は、予測期間中に7.5%のCAGRで成長すると予想されていました

調査対象のバイオ分析検査サービス市場は、予測期間中に7.5%のCAGRで成長すると予想されていました。市場の成長に起因する主な要因は、バイオマーカー、低分子などの臨床試験におけるバイオ分析試験の適用の増加です。生物分析検査サービスの需要の高まりは、感染症、HIVなどの世界的な罹患率の高まりによるものです。さらに、コロナウイルス病(COVID-19)、エボラウイルス病などの感染症の発生を制御するための政府のイニシアチブの増加は、より多くの機会を提供すると予想されます。2020年2月に発表されたWHOの報告書によると、COVID-19は80000人以上に影響を与え、世界中で2600人以上が死亡しました。したがって、生物分析評価の需要は、市場の成長に寄与することが期待されています
主な市場動向
低分子サブセグメントは、生物分析試験サービス市場で最大の市場シェアを保持すると予想されています
低分子検査サービスのサブセグメントは、ほとんどの医薬品がこのカテゴリーに分類されるため、ブランド医薬品のジェネリック版の開発において重要な役割を果たしているため、バイオ分析検査サービス市場で最大の収益シェアを保持すると予想されています。これらの生物分析試験結果は、ジェネリック医薬品の放出プロファイルをブランド医薬品の放出プロファイルとシミュレートする効率を証明する上で大きな役割を果たし、セグメントの成長につながります
一方、高分子セグメントは、予測期間中に有利な成長を目撃すると予想されます。最も速い成長は、研究活動の増加と生物製剤、ペプチド、またはアミノ酸ベースの分子の巨大なパイプラインの存在のために期待されています。さらに、高分子のバイオアナリシスには、高度な分析機器やインフラとともに、高度な技術を持つ専門家が必要です。これらは、予測期間中に市場の成長を促進する可能性が高い生物分析試験サービスプロバイダーによって容易に提供されます
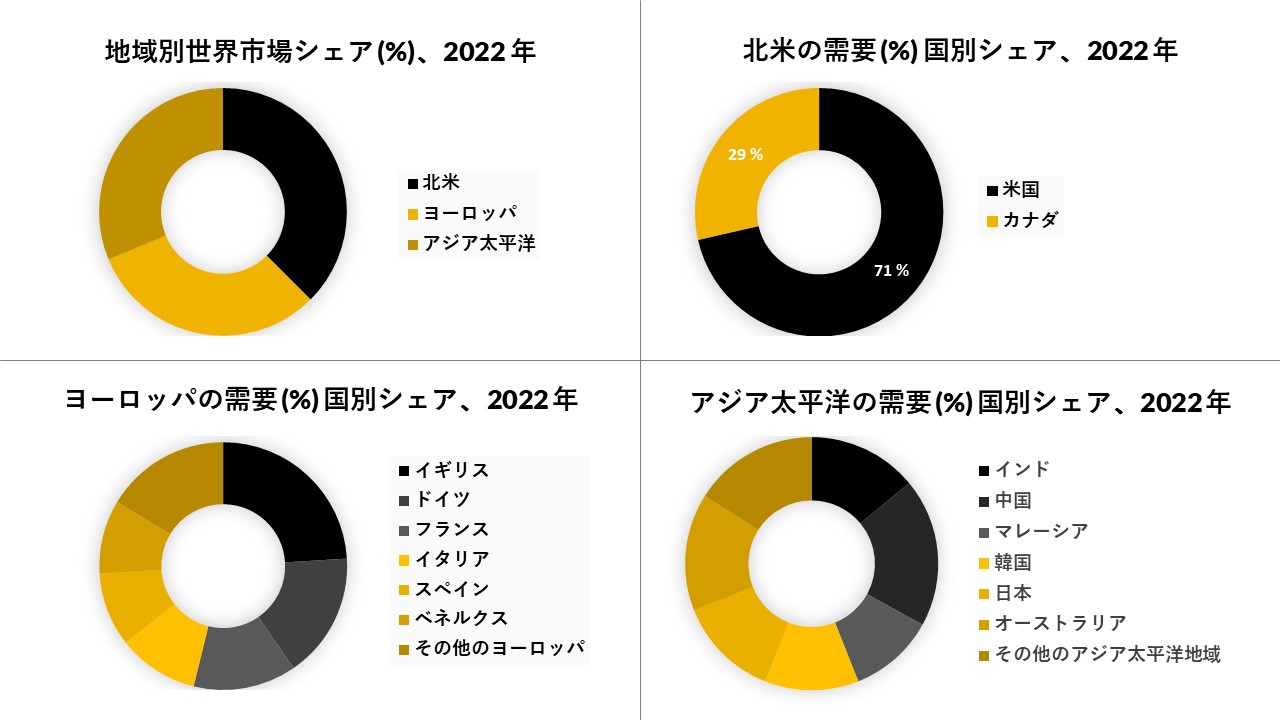
北米は市場を支配しており、予測期間
北米は、予測期間を通じてバイオ分析検査サービス市場全体を支配すると予想されています。この優位性は、進行中の膨大な量の研究活動と臨床試験を伴うバイオ分析サービスに対する需要の高まりと、この地域の市場を刺激する高度なインフラストラクチャの高い採用と相まって、北米では、米国は、様々な慢性疾患の患者プールの増加と、より多くの副作用を有する小分子の代替としてのペプチド、他の高分子治療薬の採用の増加により、最大の市場シェアを保持している。例えば、2019年の国際糖尿病連合によると、北米の約4800万人の成人が糖尿病に罹患しており、2型糖尿病の患者プールの体積の増加を止めるためのジペプチジルペプチダーゼ4(DPP-4)阻害剤、グルカゴン様ペプチド1(GLP-1)類似体などの新規(高分子化合物)ペプチド治療薬のバイオアナリシスに対する需要が高まり続けると予想されています。 この地域の市場を牽引し、予測期間中の世界市場収益の優れたシェアに貢献しています
競争環境
生物分析検査サービス市場は中程度の競争が激しく、いくつかの主要プレーヤーで構成されています。一部の企業は、さまざまな種類のサービスを提供し、メソッド開発、アッセイ検証のための新しい方法を立ち上げることによって市場の地位を拡大しており、他の企業はラボソリューションを提供しています。例えば、2020年2月、SGS SAは、バイオセーフティ研究所のセンターオブエクセレンスがコロナウイルスのGMP準拠のワクチン試験ソリューションを提供すると発表しました。2019年、バイオセーフティラボの施設は、迅速かつ高度なテストプラットフォームを使用して、新しいウイルスの脅威のテストサービスを提供する能力と能力を高めるために拡張されました。このような戦略的な拡大は、市場にプラスの影響を与えることが期待されています。現在市場を支配している企業には、SGS SA、ICON PLC、Laboratory Corporation of America Holdings、Syneos Health Inc.、Charles River Laboratories Inc.
このレポートを購入する理由:
- エクセル形式の市場予測(ME)シート
- 3ヶ月のアナリストサポート

北米(米国およびカナダ)、ラテンアメリカ(ブラジル、メキシコ、アルゼンチン、その他のラテンアメリカ)、ヨーロッパ(英国、ドイツ、フランス、イタリア、スペイン、ハンガリー、ベルギー、オランダおよびルクセンブルグ、NORDIC(フィンランド、スウェーデン、ノルウェー) 、デンマーク)、アイルランド、スイス、オーストリア、ポーランド、トルコ、ロシア、その他のヨーロッパ)、ポーランド、トルコ、ロシア、その他のヨーロッパ)、アジア太平洋(中国、インド、日本、韓国、シンガポール、インドネシア、マレーシア) 、オーストラリア、ニュージーランド、その他のアジア太平洋地域)、中東およびアフリカ(イスラエル、GCC(サウジアラビア、UAE、バーレーン、クウェート、カタール、オマーン)、北アフリカ、南アフリカ、その他の中東およびアフリカ)